![Director of National Parks [logo]](/images/dnp_90px.gif)


![Director of National Parks [logo]](/images/dnp_90px.gif) |
 |
 |
Revegetation with native grasses is often difficult. A common problem is that whilst the seed may be healthy and potentially germinable it is difficult to get it all to germinate rapidly and at the same time. This is the concept of ‘synchronicity’, which is the major theme of this report. There are two facets to synchronicity which reflect different time scales. Firstly, it is necessary to get the seed in a particular batch to ripen at the same time after harvesting. Secondly, it is desirable that germination is compressed into as short a time as possible to maximise the opportunity to utilise available soil water and to reduce predation and competition. Unlike grain crops and exotic lawn grasses which have been selected for synchronous ripening and germination it is not desirable to use this method to produce seed for revegetation with native grasses because of the need to preserve diversity in the gene pool. This study therefore examines alternative methods to facilitate revegetation with native grasses. Because of the need to make the techniques accessible and practical for all landowners only ‘low tech’ solutions have been considered. For example, the enhancement of germination by the application of gibberellic acid (GA3), which is well documented for many grass species, has been only been considered for one, rather difficult, species (Themeda triandra) because of the high cost and handling difficulties involved in the use of GA3. Similarly, the emphasis of this study has been on results (empirical), rather than on elucidating the physiological processes involved.
Synchronous germination may appear to be a relatively simple goal but on closer examination is becomes apparent that the processes involved are very complex. The propagation of grasses by seed requires the study of at least four distinct phases in the life history of the plant. These include seed ripening prior to shedding and dispersal, seed dormancy, germination and early establishment. The optimal conditions for any one of these phases may be very different to those for the others. In addition conditions during the earlier stages may influence the requirements for the later stages. For example, the conditions under which the seeds ripen on the mother plants may affect seed size, dormancy characteristics, germinability and early vigour.
Dormancy is a survival strategy that delays seed germination until conditions are favourable for growth or spreads out germination to increase the chances of at least some plants establishing. Baskin and Baskin (1998) claim that, of the various types of dormancy available to plants, grasses only exhibit physiological dormancy. This is of a type that can often be overcome by manipulating dry storage conditions (eg high temperatures, low temperatures, combinations of high and low temperature), duration of storage, injury to the seed covering layers, or the application of gibberellic acid. The recent discovery of the importance of smoke in overcoming dormancy or stimulating germination (see references in Tieu et al, 1999) may also be relevant to grasses. Genetics, the environment, and interactions between them control dormancy. The environment effect may be via its effect on the mother plant during seed maturation rather than directly on the seed. Some species produce seeds with different dormancy characteristics, either from different plants (polymorphism) or even on the same plant (heteroblasty) (Gutterman, 1993) where there are differences in germinability so that no single environmental event triggers all of the seeds to grow.
For germination another set of environmental conditions must be met. These may include, for example, temperature, light or dark, alternating temperatures, soil moisture, wetting/drying cycles, position of seed on or in the soil, and pH.
Establishment factors include rainfall patterns and distribution, topography, soil type, daylength, temperature, canopy type and degree of disturbance, competition, predation, pathogens, symbionts, and maternal effects (seed heteromorphism) (Cheplick, 1998). The presence of fungi can be either beneficial, as in mycorrhizal associations, or harmful as with pathogens. Control of fungal infections during germination experiments may therefore have unforeseen consequences.
The difficulty of elucidating the optimal environmental and biological conditions for each grass species for each phase of development may be further complicated when a solution for one part of the riddle impacts on another part. For example awns may help the seed to fall in an upright position and increase success of germination (Peart 1981) or they may locate the seed in an appropriate microsite on the soil surface (especially cracking surfaces) (Peart 1979). Hairs in the base of the caryopsis may have a similar function. If it is found that the removal of awns or seed coats helps to overcome dormancy or increases germination in laboratory conditions this may be of little value if it also reduces establishment under field conditions.
Genetic variability adds another level of complexity. Within any particular species there may be many different dormancy responses (eg Groves et al, 1982) especially if the species has a very extensive geographical range. For this reason much of the available published material can be largely irrelevant to local conditions. For practical re-vegetation it is necessary to study material indigenous to the area concerned if it is desirable to use only seed of local provenance. For the purposes of this project the seed was therefore obtained exclusively from the local district.
The species chosen for study are common within the study area, and also represent four of the six sub-families of grasses and two distinct photosynthetic pathways. The latter may be of considerable importance in choosing planting season as the ‘C3’ grasses (so called because the first products of photosynthesis have 3 carbon atoms) may be better adapted for winter growth than the ‘C4’ grasses (those in which the first products have 4 carbon atoms) and which are better adapted for summer growth.
"Germination" in this report means that the radicle (the primary root) had emerged from the seed. "Seed" is used in the broad sense, meaning the dispersal unit which falls from the plant. The seed is actually a caryopsis, which is comprised of a fruit with a pericarp fused with the testa. "Intact" seed means dispersal units cleaned of extraneous material collected as a by-product of the harvesting methods, and usually includes the awns (if present) and the seed coat. "Bare seed" refers to the caryopsis only. "Full" seed means that the caryopsis is fully developed.
Seed for all germination trials was selected by hand to ensure that full seed was used. Sometimes selection of full seed was made easier by soaking it for an hour so that the seed coat became transparent. In vitro germination tests were initially conducted in 9 cm diameter petri dishes containing 2 layers of Whatman No.1 filter paper and 3 mL solution. In early experiments the dishes were sealed with ‘Parafilm’ to reduce evaporative losses. This is a commonly used method for laboratory germination tests, but is rather time consuming as it is necessary (because of condensation) to unseal the dishes regularly to examine the seeds. Following observation of methods at a commercial seed company the method was modified and in later germination tests consisted of 1 layer of towelling on one layer of filter paper with 25 mL solution. With this amount of solution it is not necessary to top up with water for up to 60 days and the dishes do not need to be sealed. This is similar to the method described in the International Rules for Seed Testing (International Seed Testing Association, 1996).
A major problem with in vitro germination testing is contamination with fungi and bacteria. For example, in a preliminary experiment with Themeda triandra up to 52% of seeds succumbed to fungal contamination despite treating with 0.4% sodium hypochlorite (bleach) for 5 minutes before sowing. This was less of a problem with species that germinate quickly. For example Ehrharta stipoides seeds with seed coats removed germinated in only 4 days and only 1% were contaminated. Most contamination is associated with the seed coat.
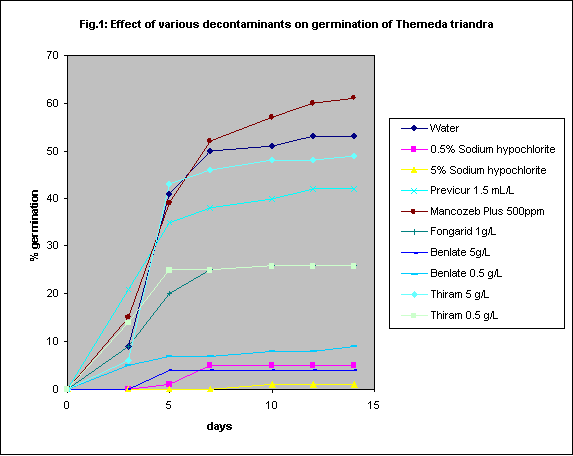
Themeda triandra seeds were used to test the efficacy of a range of chemical treatments (Fig. 1). The seeds used had previously been tested to ensure that germination rates would be adequately high (ca. 50 - 70%). The awns were removed but the seed coats were left intact. Mancozeb Plus treatment (500ppm) failed to reduce contamination. Sodium hypochlorite (5% or 0.5%) was effective in reducing contamination but seriously reduced germination as well. Thiram (5g.L-1) and Previcur (1.5 mL.L-1) reduced contamination without significantly reducing germination. In another experiment Foli-R-Fos 2000 used at 6 mL.L-1 did not significantly affect germination rate or seedling growth but did reduce contamination. As Thiram is formulated as a powder it is less easy to handle than the other chemicals in in vitro tests. However it may be advantageous to use a powder to coat seeds before field planting and would therefore be the first choice.
PPM (‘Plant Preservative Mixture’, an unspecified compound manufactured by PhytoTechnology Laboratories LCC, Washington DC, and supplied by Austratec Pty Ltd, Mt Evelyn, Victoria) was used during the second year of the project. This is a chemical compound that reduces contamination of tissue cultures. The substance was used to treat Poa labillardieri seeds either as the imbibing solution or the incubating solution or both. Imbibing in a 40 mL.L-1 solution of PPM completely controlled contamination. The seed was soaked for about 16 h. In contrast all seeds in a distilled water control became infected. Seeds imbibed in PPM and then incubated in the same solution did not become infected and produced normal shoots but failed to develop roots. A second experiment tested the effect of three different concentrations of PPM for imbibition (1, 10 or 20 mL.L-1) with incubation either in distilled water or a 1 mL.L-1 PPM solution. Contamination only occurred in seeds imbibed in the 1 mL.L-1 solution and incubated in water (35% after 36 days), and also in those imbibed as well as incubated in the 1 mL.L-1 solution of PPM (16% after 36 days). The presence of 1 mL.L-1 PPM during incubation also appeared to enhance germination (71% vs. 46%). Root development was normal in all treatments. Because of this apparent promotion of germination PPM was included in later experiments on chemical treatments to break dormancy in Themeda triandra. It was also used as a standard treatment to reduce contamination with other species.
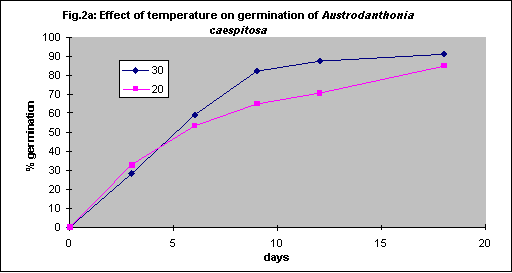
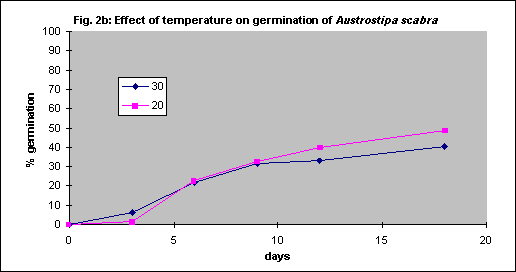
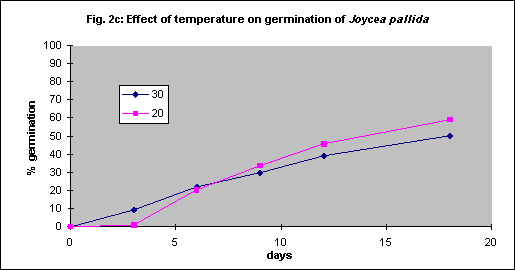
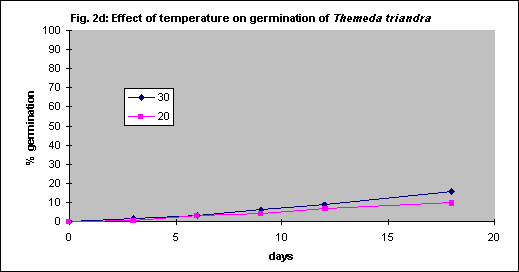
Under field conditions there is little control of soil temperature beyond choice of season for planting but for seed testing in vitro the optimum temperature was determined by experimentation. Generally germination is faster with increasing temperature, but some grasses have relatively low optima.
Four species were tested. Themeda triandra and Austrostipa scabra were tested with intact seed coats whereas bare seed was used for Joycea pallida and Austrodanthonia caespitosa. As there were no significant differences between 20 and 30° C for any of the 4 species (Figs. 2a - d) all further tests were done at 20° C for convenience.
Species selection, seed supply and seed quality
Seven species have been studied. The taxonomy of many Australian grasses has been subject to revision in recent years. To minimise confusion the names that will be used in the forthcoming Flora of Australia: Poaceae (Volumes 43 and 44) have been used in this report. The following list of species identifies the synonyms and common names that are familiar to most people. All these species are common in the ACT and adjacent areas of NSW. Details of identification, uses and responses to fertilisers and grazing are covered in ‘Native Grasses - Identification handbook for temperate Australia’ M. Mitchell 1996, Agmedia, Melbourne. Provenance information given below applies to the material used in the in vitro germination experimental program. Information on the photosynthetic pathway was determined from published sources rather than direct investigations.
Current name: Austrodanthonia caespitosa (Gaudich.)H.P. Linder
Formerly known as: Danthonia caespitosa Gaudich.
Subfamily: Arundinoideae
Common name: Wallaby grass, White top
Provenance: "Wargeila" nr. Yass, NSW
Photosynthetic pathway: C3
Current name: Austrostipa scabra subsp. falcata (Hughes) S.W.L. Jacobs & J. Everett
Subfamily: Arundinoideae
Formerly known as: Stipa scabra subsp. falcata (Hughes) Vickery, S.W.L. Jacobs & J. Everett
Common name: Spear grass
Provenance: Hume, ACT
Photosynthetic pathway: C4
Current name: Bothriochloa macra (Steud.) S.T. Blake
Subfamily: Panicoideae
Common name: Red grass, Redleg grass
Provenance: "Miowera" Paddys River, ACT, and Mt Painter, ACT, for use in field trials
Photosynthetic pathway: C4
Current name: Ehrharta stipoides Labill.
Formerly known as: Microlaena stipoides (Labill.) R.Br.
Subfamily: Bambusoideae
Common name: Meadow rice grass, weeping grass
Provenance: "Glendale" nr. Bowning, NSW
Photosynthetic pathway: C3
Current name: Joycea pallida H.P.Linder
Formerly known as: Chionochloa pallida (R.Br.) S.W.L. Jacobs
Subfamily: Arundinoideae
Common name: Red anther wallaby grass
Provenance: Australian National Botanic Gardens, Black Mountain, ACT
Photosynthetic pathway: C3
Current name: Poa labillardierei Steud.
Subfamily: Pooideae
Common name: Tussock grass
Provenance: Smokers Gap, ACT
Photosynthetic pathway: C3
Current name: Themeda triandra Forssk.
Formerly known as: Themeda australis (R.Br) Stapf.
Subfamily: Panicoideae
Common name: Kangaroo grass
Provenance: Belconnen, ACT
Photosynthetic pathway: C4
Effect of provenance on germination
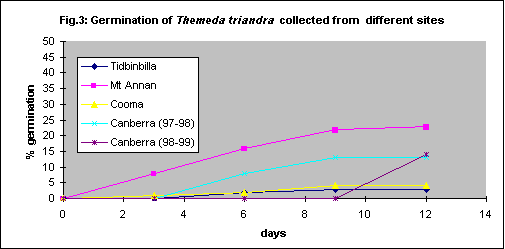
Different germination rates were found for Themeda triandra collected at four different sites. All except one collection was in January 1997 and the seeds were 2 months old when tested. The awns were removed to keep the seed in contact with the filter paper, but the seed coats were left intact. The results (Fig. 3) confirm previous findings of variability due to provenance (Groves et al, 1982). The different result for the Canberra material collected two years later may indicate the effect of the environment during the seed development phase.
Seed production in any one year is very dependent on weather conditions. For example, Table 1 shows the effect of the 1997-98 drought on the availability of seed in the local region. Much more seed was produced in the summer of 1996/97 than in the drought year 1997/98. As the best storage conditions for seed may be dependent on the length of storage required (see below) this has some practical implications. To ensure that a seed supply is available every year it will be necessary to harvest more than is needed for any one year and store the surplus in cooler temperatures.
Table 1: Effect of the 1997-98 drought on production of grass seed
|
Species |
eg. localities |
Flowers |
Seed set |
|
Austrodanthonia caespitosa |
Yass |
yes, but early |
yes |
|
Austrostipa scabra ssp. falcata |
Hume Barry Drive |
yes |
no |
|
Bothriochloa macra |
Paddys River area Canberra suburbs |
yes |
yes |
|
Ehrharta stipoides |
Yass "Tidbinbilla" |
no |
no |
|
Joycea pallida |
Aranda Black Mountain |
about 1% plants flowered |
v. low |
|
Poa labillardierei |
Smokers gap |
yes |
yes |
|
Themeda triandra |
Bruce "Tidbinbilla" |
yes |
no |
Irrigation should lead to more reliable seed supply. For example, the 100 seed weight for Themeda triandra collected on Mt Painter was 387 mg compared to 467 mg for seed collected from irrigated and fertilised plants at ANBG. However some caution should be exercised, as there may be effects on the degree of dormancy in the irrigated seed lots that could be counter productive.
Some species of grass produce many sterile flowers, which produce no seed. The seeds on native grass heads ripen gradually so many under-ripe seed units are collected at harvest and ripe seeds may have already been shed and lost. The result is a lower seed yield than would be expected in some species. In addition insects can damage seed. Hand collected seed units for all species were dissected to estimate the approximate percentage of intact, undamaged and fully-grown seed in the collections. The results are shown in Table 2.
Table 2: Seed quality of hand collected seed
|
Species |
% intact seed |
|
10% |
|
|
Austrostipa scabra |
50% |
|
Bothriochloa macra |
100% |
|
Ehrharta stipoides |
30% |
|
Joycea pallida |
10% |
|
Poa labillardierei |
26% |
|
Themeda triandra |
10% |
Similarly, a sample of Austrodanthonia caespitosa ‘seed’ collected with a mechanical harvester contained only 10% seed by weight. Only 25% of the florets contained seed. In practical terms 1 kg of raw ‘seed’ contained about 120,000 germinable seed (100g).
Later assessment of seed quality for all species except Themeda triandra and Austrostipa scabra was done by soaking the seed in water for a few hours then examining under a microscope. This makes the seed coat transparent so that the presence and size of the seed can be easily observed. It also facilitates the removal of the seed coat for germination tests where this is desirable. In the case of Ehrharta stipoides seed maturity is indicated by the seed coat changing colour from green to green/brown. Germination of green seed is about a third less than mature seed.
Themeda triandra is commonly collected with a large amount of hay and the seed units may represent only about 1% by weight of the collection. This may be true for other species too. Recently commercial seed suppliers in the local region have been developing more efficient seed harvesting and cleaning machinery. We therefore investigated the efficiency of various harvesting methods, including:
The relative performance of each is very dependent on the site and the purity of the sward and it is therefore not possible to make fair comparisons between them. However when seed harvested by any of these methods has been examined under the microscope prior to germination tests there is only minimal damage from harvesting.
For example, Ehrharta stipoides, harvested with a suction machine then cleaned by a seed merchant of all extraneous plant material other than the seed coat, was compared with uncleaned material form the same collection. The seed coat was then manually removed prior to germination testing. Germination was the same for both treatments (99% + after 7 days). There was no physical damage to the seeds from the cleaning process.
Similar results were obtained for Themeda triandra collected with a cutting harvester developed by John Betts and Tony Wilson and cleaned using a machine constructed by Tony Wilson. Five batches of 100 seeds were randomly selected and assessed for damage (Table 3).
Table 3: Effect of mechanical harvesting on seed quality
|
Seed condition |
% |
|
Undamaged full seed |
88 |
|
Callus broken off |
1 |
|
Top end of seedcoat chipped, inner seed intact |
6 |
|
Cracked or abraded seedcoat |
1 |
|
Dent or crack in coat and seed |
1 |
|
Total full (hard) seed |
96 |
|
Empty seed |
4 |
Machine harvesting therefore causes no problems with seed quality. Removal of seed coats may however be more difficult.
[ NEXT PAGE ]